 The first five of these series are named Lyman, Balmer, Paschen, Brackett, and Pfund, out of which only Balmer belongs to the visible region in the electromagnetic spectrum. In his observation, Rydberg concluded that the series of lines in the hydrogen spectrum could be described as an equation determining the wavenumber. This is known as hydrogen spectrum wavelength. For example: when the electron jumps from n = 4 to n = 1, the wavelength corresponding to it is 97 nm. This photon belongs to some wavelength, and the amount of energy radiated depends on the energy levels, which makes the wavelength different for different transitions. The electron loses the photon and jumps back to lower energy levels to stabilise the atom again, say n = 2 to n = 1. The electrons transit from lower energy levels to higher energy levels when the hydrogen atom absorbs photons say n = 1 to n = 2. The quantised electronic structure of an atom is perfectly illustrated by the hydrogen spectrum and helps us understand it better. The spectrum of hydrogen thus consists of several series of lines named after the scientists who discovered them for example, the Lyman series, the Pfund series, and many more. The H atoms get excited by this high power and possess an energy that emits electromagnetic radiation of discrete frequencies. In an experiment in which an electric discharge of high voltage is made to pass through a gaseous hydrogen molecule, the hydrogen molecules dissociate. Let’s understand why and how it radiates energy. During this process, the hydrogen atom’s electron emits EM radiation called the spectrum, which is a line spectrum. Due to natural reasons, this electron is bound to lose energy and come back to its normal state, which is a stable state. Whenever the electron of a hydrogen atom absorbs energy or radiation, it is called the excited electron, and the state is known as the excited state. 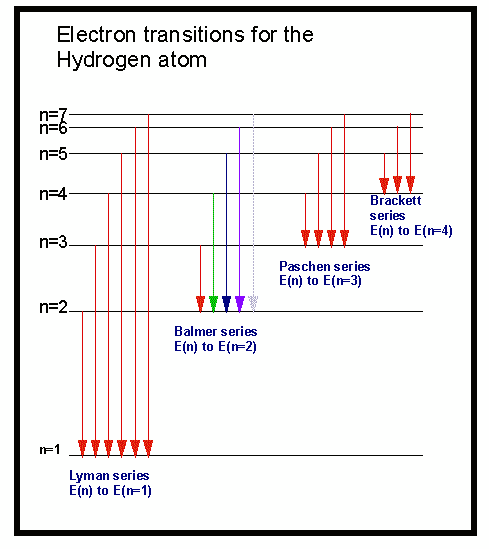
Every element in the periodic table has its properties, one of them being the emission of electromagnetic radiation. 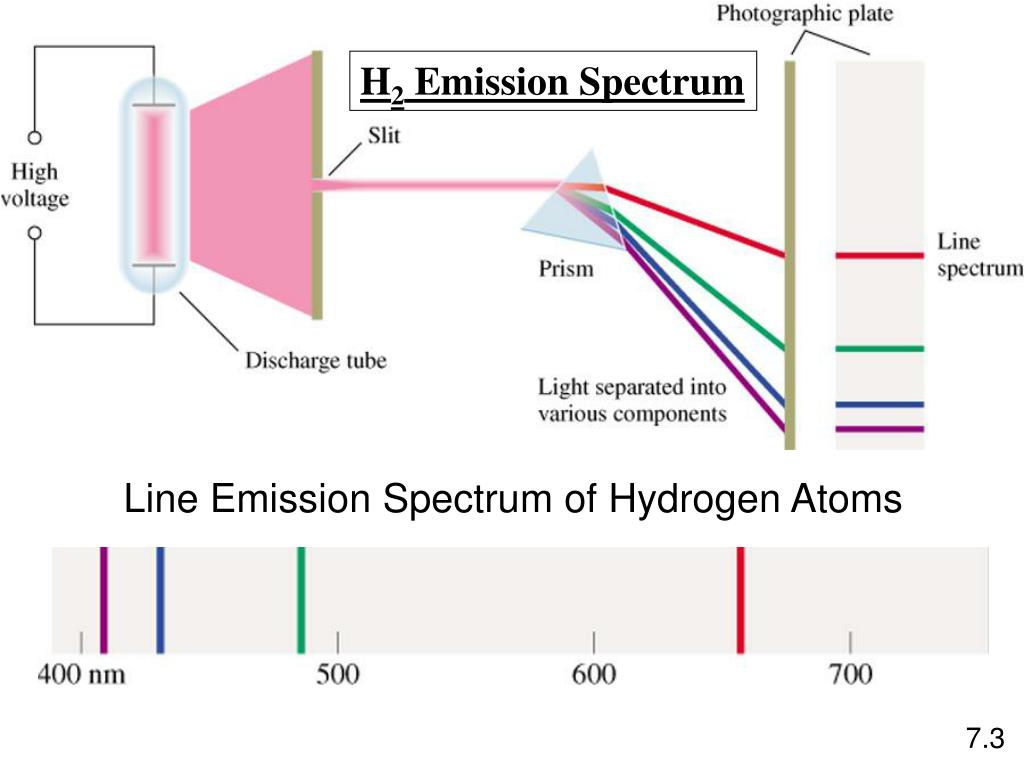
It is non-metal and consists of one electron and one proton. Hydrogen is the very first element of Henry Moseley’s modern periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
